Development of Medical and Healthcare Devices
Full-cycle development of Class I, II, and III medical electronics.
From initial hardware architecture to IEC 62304-compliant firmware. See what we can do.
We work with innovators and Fortune 500 technology leaders
Precision Engineering
for Life-Critical Medical Devices

IEC 60601
IEC 62304
IEC 82304-1
and more
How We Can Help?
Why Should You Work With Us?
With a deep technical background and a history of successful FreeBSD contributions, we provide end-to-end solutions for businesses looking to leverage FreeBSD’s capabilities.
Sample Healthcare Development Roadmap
Are you ready to entrust us with medical devices development?
Here’s what you can expect.
-
Discovery & Risk Analysis
Defining intended use and Class (ISO 14971).
-
Architecture & MVP
Hardware block diagrams and core firmware.
-
Verification & Validation
Rigorous unit testing and system integration.
-
Regulatory Support
Compiling the Technical File / DHF (Design History File).
-
Manufacturing Transfer
Moving from prototype to mass production.
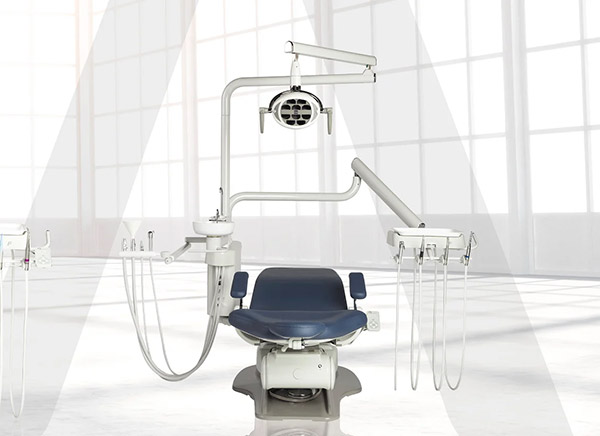
Case Study: Development of a Modern Dental Chair Control Board with IoT Capabilities
Here's what working with Conclusive Engineering looks like in practice.
Reviews and Testimonials
Customers value our services and here's proof.
Updates
& Communication
We understand that good communication is the key to success. That’s why our engineers always stay in touch with your team to discuss the project.
We usually do the following for our clients:
- dedicated Slack channel,
- dedicated project supervisor,
- regular project updates,
- ability to work in the client’s time zone,
- on-site visits,
- and more.
Cooperation
Are you interested in working with Conclusive Engineering? We can offer different payment options, such as time & material, fixed price, or hybrid alternatives.
Read more about our cooperation schemes
Talk to Conclusive Engineering Experts
Submit your project details and a Conclusive Engineering expert will contact you soon to discuss how we can support your project.
Trusted partner by leading tech companies:
“We found that they were very resourceful; they suggested improvements even though they didn't have expertise in our specific industry, which ultimately resulted in a product that exceeded our initial requirements."
Robert Young
VP of R&D, Dental Products & Services Company
Talk to Conclusive Engineering Experts
Submit your project details and a Conclusive Engineering expert will contact you soon to discuss how we can support your project.
Yes, we maintain full traceability.
You do. Full IPR transfer is our standard.
Yes, we specialize in hardware optimization.